Water Chemistry Monitoring
Test physical and chemical characteristics of water to determine pollution sources and long-term trends in water quality. Six variables are measured with a customized test kit, and results can be compared with water quality standards that define conditions for healthy waterbodies.
Supplies
Alabama Water Watch only accepts water chemistry data that has been collected with the approved equipment described below.

LaMotte Alabama Water Quality Monitoring Kit
Call LaMotte Company’s Customer Service department at 1-800-344-3100 to place your orders. LaMotte does not have an online purchasing option. The hours of operation are 8:15 AM to 5:00 PM EST, Monday through Friday. When placing an order tell them that you are a volunteer with the Alabama Water Watch Program, and use the Catalog numbers below to ensure you receive the correct product. LaMotte accepts most major credit cards. Some organizations qualify for net 30 day open account terms, or to pay with purchase orders. Contact LaMotte for additional information.
Alabama Water Quality Monitoring Kit catalog No. 9844-02 – $359.00
Alabama Water Quality Monitoring Kit with Secchi Disk catalog No. 9845-02 – $552.00
Full Refill – Alabama Water Quality Monitoring Kit, catalog No. R-9844-01 – $137.70
Prices as of October 2023
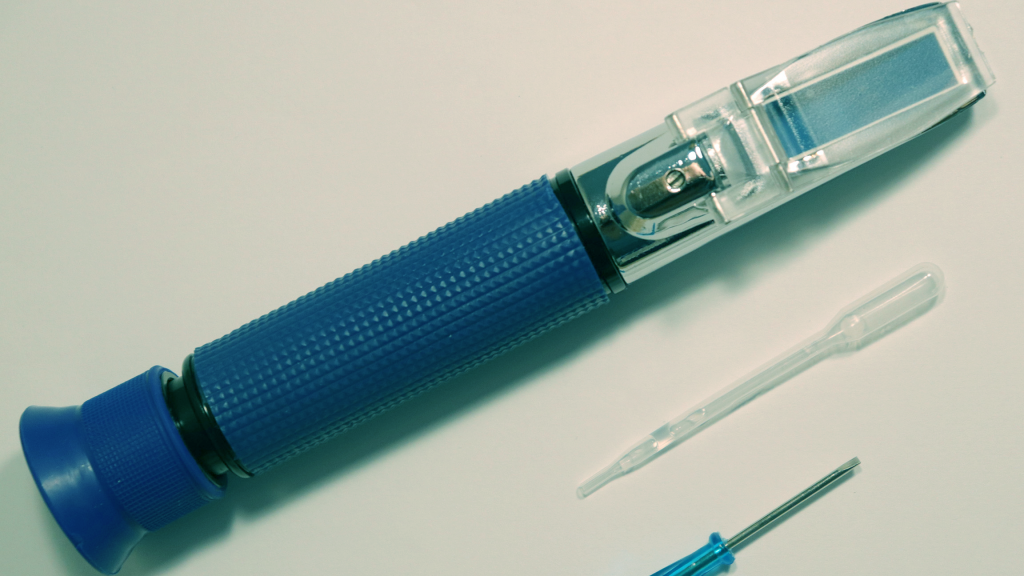
Refractometer
Monitoring in brackish or saltwater sites may require the use of a refractometer to measure salinity. AWW recommends the following model.
Agriculture Solutions LLC Salinity Refractometer – for Coastal Groups (1.000/1.070), catalog No. SKU AGT-SSA0010 – $60.00
Order by calling Agriculture Solutions LLC at 1-888-683-8291 or through the Agriculture Solutions LLC Website.

Secchi Disc
Volunteers who monitor lakes, ponds, or other deep waters may use a Secchi Disc to measure water clarity. AWW recommends the following model.
Forestry Suppliers Fieldmaster® Student Secchi Disc – $44.75

